F2 Labs provides complete CE mark testing and compliance services to help you meet European Union requirements for product safety, EMC, and performance. Our expert guidance simplifies the CE certification process and supports fast, successful market entry.
CE Mark Testing & Certification
The CE mark is a mandatory European marking for certain product groups to indicate conformity with the essential requirements set out in European Directives. In order to use the CE mark on a product, the manufacturer must draw up an EU declaration of conformity (DoC) in which the manufacturer attests to conformity with all relevant New Approach Directives (NADs) and takes sole legal responsibility. In some instances, a NAD may require a Notified Body to issue a Certificate of EU declaration of conformity (DoC) Conformity in order to verify the performance of the product or constancy of the production process (Factory Production Control, for example). F2 Labs can assist you in identifying the requirements for CE certification. We have a staff of experts experienced in the CE mark who are ready to assist you throughout the process of CE testing and CE certification. With more than 20 years of experience in CE testing and evaluation, F2 Labs has helped many businesses successfully enter into the European and global markets with CE certification.We, at our CE Mark Testing Lab test products to the following directives:
- Radio Equipment Directive 2014/53/EU (RED), formerly R&TTE Directive
- EMC Directive 2014/30/EU (formerly Directive 2004/108/EC)
- (ATEX) Directive 2014/34/EU (formerly Directive 94/9/EC)
- General Product Safety Regulation 2023/988
- Low Voltage Directive 2014/35/EU (formerly Directive 2006/95/EC)
- Machinery Directive 2006/42/EC
- Medical Devices 93/42/EEC
Steps to Obtaining the CE Mark
F2 Labs will walk you through each step of the CE testing certification process:
- Identify the directive(s) and harmonized standards applicable to the product
- Verify the product-specific requirements
- Identify whether a Notified body approval is required
- Test the product to check its conformity
- Compile and keep available the required technical documentation
- Affixation of the CE mark to your product and complete an EU declaration of conformity
The EU declaration of conformity (DoC) must include: manufacturer’s details (name, address, etc.); description of the product; List of EU Directives to which the product complies; list of relevant EN standards used; must be signed and dated by an authorized representative of the company placing the product on the European Market, and must be translated into the native language of the country in which it will operate.
The CE mark is covered by Council Decision 93/465/EC. Annex B(d) provides the following guidelines:
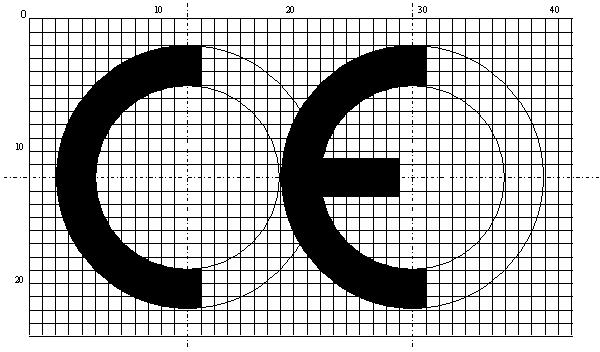
- The CE conformity marking must consist of the initials ‘CE’ taking the following form:
- If the CE conformity marking is reduced or enlarged the proportions must be kept the same.
- Where the directive concerned does not state specific dimensions, the CE marking must have a height of at least 5 mm.
- The CE marking must be affixed to the product or to its data plate. However, if that is not possible or warranted on account of the nature of the product, it must be affixed to the packaging, if any, and to the accompanying documents, where the directive concerned provides for such documents.
- The CE marking must be affixed visibly, legibly, and indelibly.
Helpful Links:
CE Mark Testing Lab
EU Directive Testing – F2 Labs has more than 20 years of experience in assisting our clients with CE testing certification, European product compliance evaluation and testing to the applicable EN standards under the EU Directives so that they can obtain the CE mark for their product(s).
EMC Directive 2014/30/EU (formerly Directive 2004/108/EC) -Our CE Mark Testing Lab can perform Electromagnetic Compatibility (EMC/EMI) testing of your product at our facility or on-site at your facility.
Low Voltage Directive 2014/35/EU (formerly Directive 2006/95/EC) – F2 Labs can provide the testing services you need in order to prove that your product is compliant with the Low Voltage Directive.
Machinery Directive 2006/42/EC Evaluation & Testing – F2 Labs can perform the evaluation and CE certification testing required by the Machinery Directive at our facility or yours.
EU Authorized Representative – F2 Labs offers can serve as your Authorized EU Representative serving as the contact point.
Medical Device Directive 93/42/EEC – Manufacturers must comply with the medical device directive in order to sell their medical products into Europe.
Radio Equipment Directive 2014/53/EU (RED), formerly R&TTE Directive – F2 Labs can assist you with your Radio Equipment Directive 2014/53/EU (RED), formerly R&TTE Directive, testing needs.
General Product Safety Regulation 2023/988 – Although this Directive does not involve CE marking the product, it may be a requirement for your European compliance project. Our CE Mark Testing Lab can assist you with testing required for compliance with the General Product Safety Directive.
WEEE Directive 2012/19/EU Testing – F2 Labs sales and engineering staff is always available to assist you with your European projects. We can talk with you regarding these WEEE disposal laws and offer our best guidance.
RoHS Directive 2011/65/EU – RoHS is a CE marking Directive and if your product is within its scope then it must carry a CE marking and be referenced on an accompanying declaration of conformity.
Food Contact Materials Regulation (EC) 1935/2004 – The European Union has specific legislation for equipment and products that come into contact with food and it has its own mark. F2 Labs can assist you with the compliance requirements for this regulation.
What is the CE Mark?
The CE mark is a mandatory European marking that indicates a product conforms to the essential requirements set out in European Directives. It demonstrates compliance with EU safety, health, and environmental protection standards.
What Products Require the CE Mark?
Certain product groups within the European Union require the CE mark.
Products That Require the CE Mark:
- Radio Equipment Directive
- EMC Directive
- ATEX Directive
- Low Voltage Directive
- Machinery Directive
- Medical Device Directive
If your product falls under one of these categories, it must comply with the relevant directives to carry the CE mark.
What is an EU Declaration of Conformity (DoC)?
The EU Declaration of Conformity (DoC) is a document in which the manufacturer declares that their product meets all relevant New Approach Directives (NADs). This document:
-
Is a Mandatory Step for affixing the CE mark.
- Confirms that the manufacturer assumes Full Legal Responsibility for Compliance.
Steps to Obtaining the CE Mark
The CE certification process involves the following steps:
STEP 1 – Identify applicable directives and standards for your product.
STEP 2 – Verify product-specific requirements to meet directive standards.
STEP 3 – Determine if Notified Body approval is necessary.
STEP 4 – Test the product to confirm conformity.
STEP 5 – Compile technical documentation to demonstrate compliance.
STEP 6 – Affix the CE mark visibly and legibly to the product or its packaging.
STEP 7 – Complete the EU Declaration of Conformity (DoC).
When is a Notified Body Required for CE Certification?
The CE mark must be:
A Notified Body is required when certain New Approach Directives (NADs) demand third-party verification. This might include assessments such as:
- Factory Production Control
- Performance testing or consistency checks for specific products
- If your product falls under these requirements, working with a Notified Body ensures compliance.
How Should the CE Mark Be Affixed?
The CE mark must be:
- Visible, legible, and indelible on the product or its data plate.
- Affixed to the packaging or accompanying documents if not feasible on the product.
- Maintained with proportions specified in the directive.
- At least 5mm in height if no specific dimensions are stated.
What Should Be Included in the EU Declaration of Conformity?
The EU Declaration of Conformity (DoC) must include:
- Manufacturer’s details (name and address)
- Product description
- List of relevant EU Directives and EN standards complied with
- Authorized representative’s signature and date
- Translation into the local language of the country of operation
Check the specific directives applicable to your product for exact requirements.
How Can F2 Labs Assist with CE Certification?
F2 Labs specializes in CE testing and evaluation services, helping businesses enter European and global markets seamlessly. With over 205 years of combined experience, F2 Labs can:
- Identify applicable requirements for your product.
- Conduct product testing to ensure compliance.
- Guide you through every step of the CE certification process efficiently.
For expert assistance, trust F2 Labs to help your business achieve CE compliance with confidence.