- By Keith Cooper
- No Comments
Your Router Might Be a Spy: FCC Cracks Down on Foreign-Made Devices
In a move that sounds like it came straight out of a cybersecurity thriller, the FCC has decided that your humble home router might
- By Keith Cooper
- No Comments
FDA Gives Its Standards List a Tune-Up (Because Even Regulations Need Updates)
The U.S. Food and Drug Administration (FDA) has rolled out its latest refresh of the Recognized Consensus Standards List, and for medical device manufacturers,
- By Keith Cooper
- No Comments
Wellness Meets Regulation (But With a Smile): An Industry Insider’s Take on FDA General Wellness Policy
Every wellness startup eventually asks the same nervous question: “Are we about to become an FDA-regulated medical device… or can we keep building?” If
- By Keith Cooper
- No Comments
Proposed Updates to MDR and IVDR for Europe
EU Wants Less Red Tape and More Medical Tech Magic (With Fewer Forms to Fill!) If you thought medical device rules in Europe were
- By Keith Cooper
- No Comments
NFPA 790 vs. NFPA 791: A Field Evaluator’s Guide to This Dynamic Duo
If you’ve ever been summoned to inspect an unfamiliar piece of equipment in the field—armed with nothing but a safety vest, questionable optimism, and
- By Keith Cooper
- No Comments
Why Does EMC Testing of a Medical Device to IEC 60601-1-2, AIM 7351731, and/or Common EM Emitters Testing Have to be Conducted in Both Charging and Battery Modes? (Even When the Device Is Basically Just Napping on the Charger)
If you’ve ever prepped a medical device for a regulatory review, such as an FDA 510K submission, you’ve probably hit that moment where you
- By Peter Jolles
- No Comments
Is My Specially Designed Equipment in the Scope of RoHS?
We assist clients with their CE marking obligations daily at F2 Labs. This often means that we get into the weeds of the Directives
- By Peter Jolles
- No Comments
We Offer CE Marking Training For Your Organization
F2 Labs offers onsite, and occasionally remote, CE marking Training for your company. Recently, I conducted our CE marking Training for a client of
- By Keith Cooper
- No Comments
So You Need to Field Evaluate Your Equipment – What You Need to Know
If an Authority Having Jurisdiction (AHJ) or electrical inspector has ever informed you that you have failed your inspection or that you require a
- By Keith Cooper
- No Comments
Corrosion | Salt Fog Testing: Like taking your product to the beach
Imagine this: your brand-new, bright, glistening product steps into a chamber. The door closes. Inside, it’s not the day at the spa it expected
- By Keith Cooper
- No Comments
Essential Performance: What It Is, Why It Matters, and When You Can Say “No Thanks”
Let’s talk about Essential Performance—the term that sounds like it belongs in a motivational poster but is actually a cornerstone of medical device compliance.
- By Keith Cooper
- No Comments
Adapting to Evolving U.S.-China Regulatory Dynamics
Medical device companies operating across U.S. and Chinese markets are facing a rapidly shifting regulatory environment. As geopolitical tensions between the two nations intensify,
- By Keith Cooper
- No Comments
REACH/RoHS and Continuing Compliance
* Until August 15th, if you mention this blog post we will offer special pricing (20% off) your full REACH and RoHS evaluation with
- By Clare Wolffe
- No Comments
Hexavalent Chromium Faces EU Ban Under New Regulatory Shift
ECHA has proposed a new EU-wide restriction to protect the public from the effects of hexavalent chromium, Cr(Vi). ECHA assessed the risks and found
- By Clare Wolffe
- No Comments
Unregulated Certificates Warning and The Implications Under EU Law
Recently, the European Commission updated some of the guidance regarding the EMC, ATEX, and Low Voltage Directives (and others) to include a warning about
- By Peter Jolles
- No Comments
Is Machinery in Scope of the RoHS Directive?
“My machine is industrial machinery, do I have to comply with RoHS for CE marking?” We are frequently asked this question. The answer is
- By Keith Cooper
- No Comments
Taming the EMC Beast: A Guide to Pre-Compliance Testing
Ah, the thrilling world of electromagnetic compatibility (EMC) testing! If you’ve ever dreamed of spending quality time with spectrum analyzers and line impedance stabilization
- By Clare Wolffe
- No Comments
Prop 65 Alert: New Chemical Found in Plastics, Packaging and Paints
In January, a new chemical was added to the California Proposition 65 list. Vinyl Acetate was found to cause cancer and has been detected
- By Clare Wolffe
- No Comments
How to Navigate California Proposition 65
Almost anything being sold in California may require a Proposition 65 warning. Proposition 65 is a California law titled the Safe Drinking Water and
- By Clare Wolffe
- No Comments
Breaking Down California’s New Proposition 65 Labeling Guidelines
The Office of Environmental Health Hazard Assessment published regulatory changes to the short-form warning label requirements in 2025. The changes will affect the short-form
- By Peter Jolles
- No Comments
ECHA Adds Five New Substances to SVHC List
The European Chemical Agency, ECHA, added five new substances to the list of Substances of Very High Concern (SVHC) overnight. The EU’s SVHC list
- By Peter Jolles
- No Comments
Does Customs Really Check For RoHS in The EU?
Yes. I get an email every week from the European Commission’s Safety Gate that lists some of the most dangerous products that were recently
- By Keith Cooper
- No Comments
Embracing AI in Healthcare: The FDA’s Guidance on Good Machine Learning Practice
Embracing the Future: The FDA’s Guidance on AI in Medical Devices As we enter an exciting era of technological advancement, the role of artificial
- By Keith Cooper
- No Comments
What is an NRTL Field Label and What do I Need to Know About Them?
If an Authority Having Jurisdiction (AHJ) or electrical inspector has ever informed you that you have failed your inspection or that you require a
- By Keith Cooper
- No Comments
The FDA and Common Electromagnetic (EM) Emitters – What You Need to Know
The FDA, per their Guidance Document published June 6, 2022, requires that medical device manufacturers be able to show that their product is immune
- By Cristina Akinwale
- No Comments
Easy Approach to Essential Performance in Medical Devices for FDA
F2 Labs has many conversations every week with medical device manufacturers who are struggling to define Essential Performance for their device. From our experience,
- By F2 IT
- No Comments
Navigating CE Certification for Entry Into the European Market
As both established and startup consumer electronics manufacturers look to expand their market reach, the European Union presents an enticing, lucrative growth opportunity. Entry
- By Keith Cooper
- No Comments
Reese’s Law – What You Need To Know
Reese’s Law was signed by President Biden on August 16, 2022 and as of March 19, 2024 all products that incorporate button batteries or
- By Peter Jolles
- No Comments
The Machinery Regulation(EU)2023/1230 vs. Machinery Directive 2006/42/EC – What Is New?
The Machinery Regulation (EU)2023/1230 was published in 2023 but does not formally replace the Machinery Directive 2006/42/EC until 2027. F2 Labs performed a deep
- By Peter Jolles
- No Comments
Hazardous Location Evaluations – ‘Behind the Scenes’ Details about Intrinsic Safety
Today, I read an email from our HazLoc engineering department that I think is extremely useful not only for F2 Labs personnel, but also
- By Keith Cooper
- No Comments
FDA Advises Medical Device Manufacturers to be Vigilant and Validate Data
The U.S. Food and Drug Administration recently sent out an Open Letter to Industry to remind manufacturers of medical devices that they should always
- By Peter Jolles
- No Comments
Helpful Guide if you are new to RoHS, REACH, WFD, & SCIP
Last June I had a discussion with an F2 Labs customer about the confusion of RoHS and REACH, and that she did not have
- By Keith Cooper
- No Comments
WPT for EVs and the Effect on AM Radio Broadcasts
As more and more electric vehicles (EVs) continue to be utilized both by the public and by government agencies to help reduce our carbon
- By Peter Jolles
- No Comments
Complying with REACH & RoHS – and the Need for Continued Review
We evaluate your device and keep it on autopilot, painlessly. The RoHS Directive 2011/65/EU+(EU)2015/863 started life in 2002 as the RoHS Directive 2002/95/EC. It
- By Peter Jolles
- No Comments
Installing New Pickups from Seymour Duncan
A benefit of working at F2 Labs is that I get to see new and innovative products, straight from the bench of the inventor,
- By Peter Jolles
- No Comments
New OSHA Requirements for Batteries in the US.
F2 Labs sent a notification to most of our customers regarding some new requirements pertaining to battery cells and battery packs in listed products.
- By Peter Jolles
- No Comments
RoHS Compliance – Suggestions From a Pro
MY SUGGESTIONS AFTER REVIEWING HUNDREDS OF PRODUCTS F2 Labs assists our customers with compliance issues for many markets across the world. We test equipment
- By Peter Jolles
- No Comments
The Machinery Directive is applicable to many Non-Electric Devices
A lot of our efforts at F2 labs are spent determining the applicability of different regulations to our customers’ products. We help you find
- By Peter Jolles
- No Comments
The EU REACH Regulation and US manufacturers… here is what you need to know.
F2 Labs assists manufacturers around the world with compliance obligations. Historically we have been an accredited test laboratory for electrical, electro-mechanical, EMC, and radio
- By Keith Cooper
- No Comments
What Now?
A comprehensive guide into the listing standards that are required after testing to compliance marks.
- By Peter Jolles
- No Comments
Compliance for Electric Bicycles, E-Bikes, and the EU
Understanding e-bike compliance under the scope of the EU machinery, electromagnetic compatibility, and restriction of hazardous substances directives.
- By Keith Cooper
- No Comments
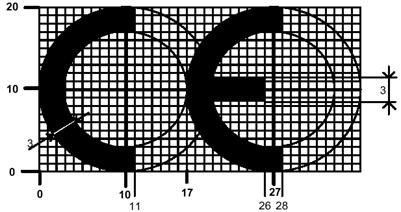
What is a CE marking? Does My Product Need One?
The CE mark is a mandatory European marking for certain product groups to indicate conformity with the essential requirements set out in European Directives. To
- By F2 IT
- No Comments
What is the UKCA Marking?
The UK has left the EU and new rules concerning the transition for most CE products to UKCA products began in January 2021. The
- By Peter Jolles
- No Comments
FDA approval for a Respiratory Monitor used in COVID19 Treatment – Tested at F2 Labs
It is safe to say that COVID-19 has affected everyone in the world. F2 Labs immediately joined the fight and offered pro-bono services to
- By Peter Jolles
- No Comments
RoHS 2011/65/EU and (EU)2015/863 – What is the difference?
Understanding the requirements for compliance to RoHS 2011/65/EU is not difficult (and we can help you), but ignoring them can get your product stopped
- By Peter Jolles
- No Comments
My product connects by USB, why do I need to comply with the Low Voltage Directive?
The question in the title of this blog article is frequently asked by our customers. Let’s break it down. See the LVD 2014/35/EU, Article
- By Peter Jolles
- No Comments
Rapid Alerts and RoHS in the European Union
I get an email from RAPEX every Friday morning that details products stopped in customs in the EU for various issues. These are not
- By Peter Jolles
- No Comments
Here is why it is important to select the right Power Supply
Most of our customers must comply with the EMC Directive 2014/30/EU prior to application of a CE marking to their products. This is because
- By Peter Jolles
- No Comments
Is the CE marking still valid now that Brexit has happened?
Yes, for now. CE marking will be accepted until at least the end of 2020 and then the UKCA mark will be the replacement
- By Peter Jolles
- No Comments
Fender Guitars and EU compliance, interesting read
In the interest of full disclosure, I own a Fender American Professional Telecaster, in butterscotch blonde of course, and a Fender Custom ’57 Champ
- By Peter Jolles
- No Comments
Machinery Directive 2006/42/EC – The text and the harmonized EN standards
Complying with any EU CE marking Directive will require a trip to one of many European Commission websites to assist with the process. They
- By Peter Jolles
- No Comments
EN 60950-1 is still acceptable for EU compliance… for now.
We are starting to feel some effects of the upcoming replacement of EN 60950-1 by EN 62368-1. This is causing some confusion in the
- By Peter Jolles
- No Comments
RoHS 3 and the new (EU)2015/863 Substances
Now that we have been under the ‘new’ EU RoHS for a few months I thought it would be a good time to write
- By Peter Jolles
- No Comments
F2 sorts out Compliance Requirements for you
Manufacturers contact F2 Labs from around the world for assistance with EU regulations. While F2 Labs primarily focuses on electro-mechanical devices, including medical devices,
- By Peter Jolles
- No Comments
Ronin Revolution earns EC-Type Examination Certificate
Ronin Revolution Corporation worked with F2 Labs to evaluate and test the Ronin Lift for compliance with the relevant CE marking Directives required to
- By Peter Jolles
- No Comments
CE marking – Equipment is stopped in French customs, what now?
The sales department at F2 Labs fields phone calls from primarily US manufacturers. We have heard every scenario imaginable as pertains CE marking, from,
- By Peter Jolles
- No Comments
Guidance For Wire Colors in Machines in the EU
We help manufacturers every day with the task of CE marking their equipment for shipment to the EU. Very often we are asked questions
- By Peter Jolles
- No Comments
RoHS 2011/65/EU – New Restrictions July 2019
RoHS 2011/65/EU presently requires compliance for electrical and electronic equipment to these six substances indicated in Annex II: We note that RoHS 2011/65/EU was
- By Peter Jolles
- No Comments
EN 62368-1:2014 will replace EN 60950-1 and EN 60065
EN 62368-1:2014 will replace two main EN standards on December 20, 2020. A year or two years ago this seemed a long way off.
- By Peter Jolles
- No Comments
Changes to the Machinery Directive 2006/42/EC
Some pretty drastic changes to the Machinery Directive 2006/42/EC were published on the European Commission’s Machinery Directive page on March 18, 2019. Most noticeably,
- By Peter Jolles
- No Comments
New RoHS standard – EN 63000:2018
CENELEC has published a new RoHS standard, announcing it, here. The new standard is EN ISO 63000:2018 Technical Documentation for the assessment of electrical
- By Peter Jolles
- No Comments
What is the Critical Component List?
F2 Labs performs safety evaluations & testing, EMC testing, and consulting for various compliance related issues faced by our customers. We open safety evaluations
- By Peter Jolles
- No Comments
RoHS 2011/65/EU and a Tale of Three Guitars
The world has many good guitar manufacturers but it is commonly accepted that the very best guitars are made in the United States. Few
- By Peter Jolles
- No Comments
Harmonized Standards and Why They Are Important
The main electrical safety standard for machinery, EN 60204-1, was revised by CENELEC and published on September 14, 2018. The currently harmonized version, EN
- By Peter Jolles
- No Comments
The Machinery Directive 2006/42/EC and Interchangeable Equipment
As usual, I opened the RAPEX Weekly Report that delivers to my inbox every Friday morning. Scrolling through, I reviewed three cases involving saw
- By Peter Jolles
- No Comments
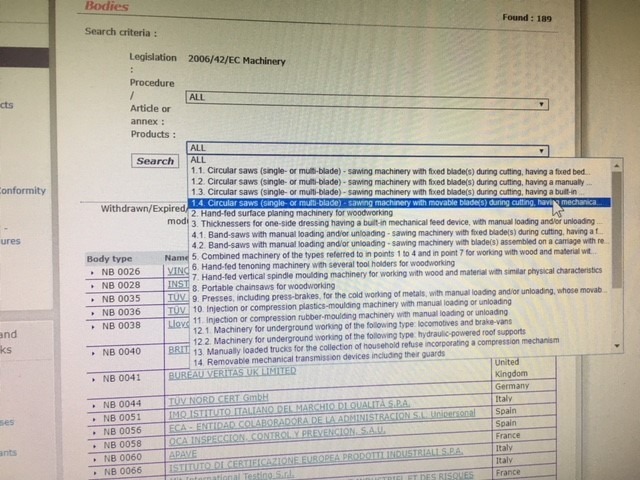
The Machinery Directive 2006/42/EC and Notified Bodies
What is a Notified Body and what does it have to do with the Machinery Directive 2006/42/EC? In most cases presented to us, absolutely
- By Peter Jolles
- No Comments
The Ambiguity of the CE Marking Process
Yesterday I posted an article pertaining to last week’s RAPEX report and the scope of the Low Voltage Directive 2014/35/EU. Today I reviewed this
- By Peter Jolles
- No Comments
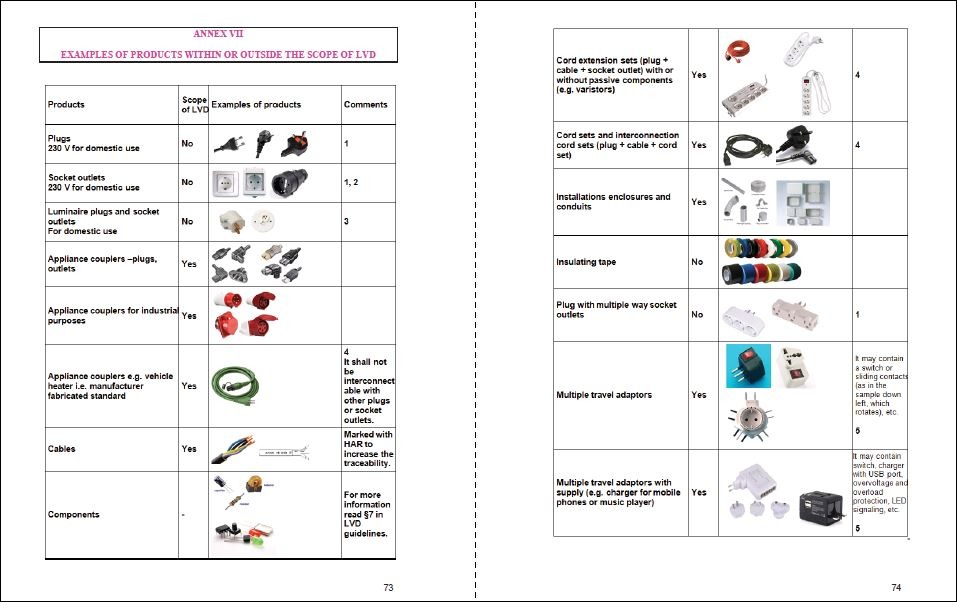
My product is DC powered. Why do I need to comply with the Low Voltage Directive?
“F2 Labs is not the EU police and I do not know any EU police.” I say that very often to customers when I
- By Peter Jolles
- No Comments
Automatic Bar Controls, Inc. – Pizza Sauce Dispenser
F2 Labs and Automatic Bar Controls, Inc. have collaborated on many compliance projects over the years and so we are pretty familiar with some
- By Peter Jolles
- No Comments
REACH Regulation (EC) No 1907/2006 – Article 7, Annex VII, and practical considerations for manufacturers – the CRASH COURSE
One possible cause for an audible “gulp” coming from the reader of a specifications sheet is coming across a requirement to comply, vaguely, with
- By Peter Jolles
- No Comments
Partly completed machinery and the requirement for Assembly Instructions
Manufacturers who build parts of machines or incomplete machinery (for example: no controls) must be aware that their equipment must not carry a CE
- By Peter Jolles
- No Comments
CE marking Onsite Consulting and Training
F2 Labs is able to send a CE marking expert to your facility to provide overview training to your business – sales and engineering.
- By Peter Jolles
- No Comments
Spare Parts and the RoHS Directive
The RoHS Directive 2011/65/EU was amended in November 2017 by Directive (EU) 2017/2102 with some administrative clarifications. I found one item particularly interesting: Recital
- By Peter Jolles
- No Comments
RoHS 2011/65/EU and Machinery
F2 Labs assists many manufacturers, internationally, with CE marking requirements. Regarding CE marking, primarily we test and evaluate electro-mechanical equipment for compliance with electrical
- By Peter Jolles
- No Comments
FCC – New Labeling and Manual Requirements
The FCC released new guidance and procedures for manufacturers to show compliance with FCC regulations in June 2017. Some of the changes are in the
- By Peter Jolles
- No Comments
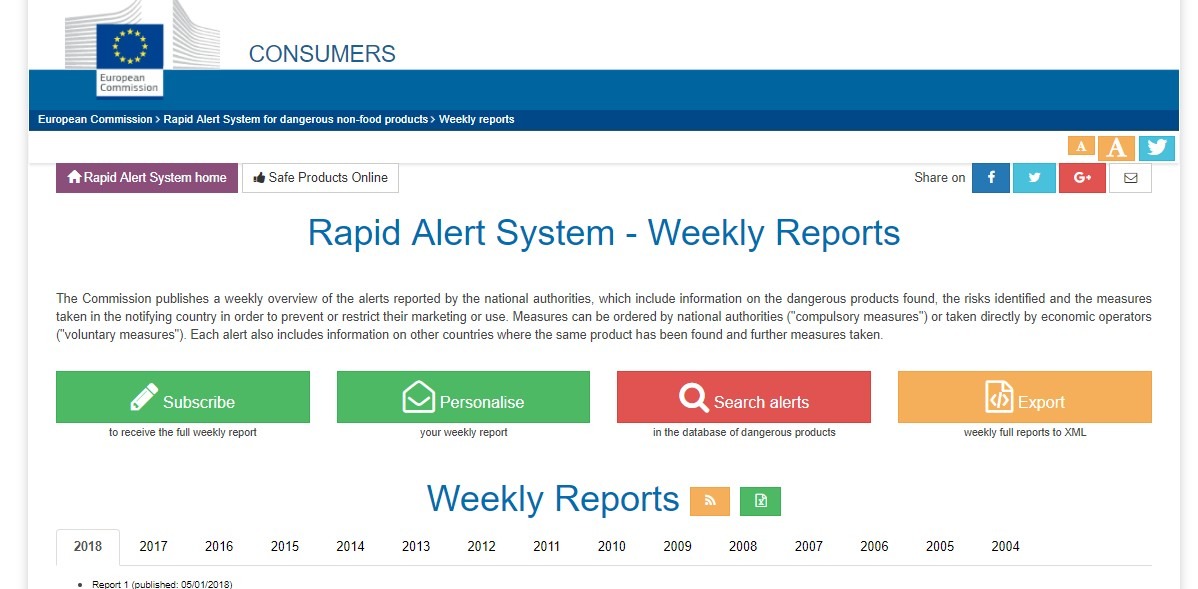
EU’s Rapid Alert System for dangerous non-food products
The EU has a direct conduit for market surveillance authorities to communicate information about non-compliant products directly between 31 EU member nations and the
- By Peter Jolles
- No Comments
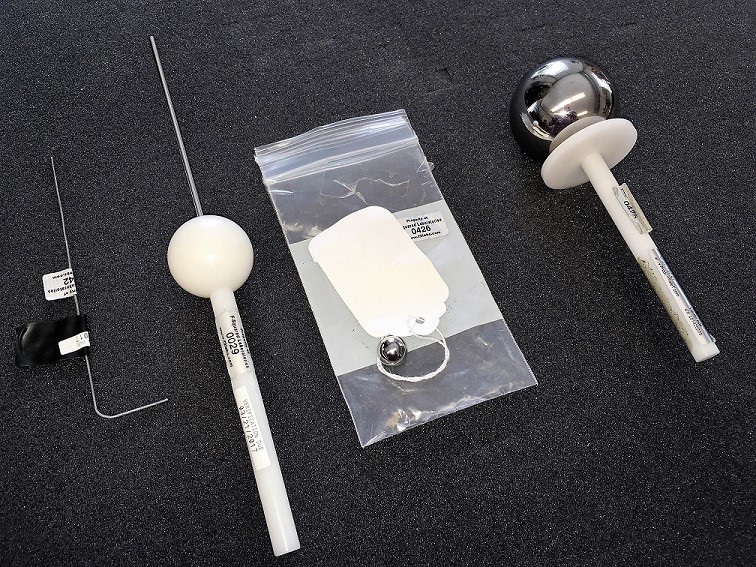
IP69 and IP69K – What Are They?
We are presented with a request for quote for IP69K testing on a regular basis at F2 Labs. Usually my first reply to the
- By Peter Jolles
- No Comments
RoHS 2011/65/EU, EN 50581:2012, and clause 4.3.3.
Manufacturers that send equipment to the EU with a CE marking must understand that a CE marking is a blanket claim by the manufacturer.
- By Peter Jolles
- No Comments
Safety Fences and the Machinery Directive 2006/42/EC
While reading the new Guide to application of the Machinery Directive 2006/42/EC, Edition 2.1, I was interested to see that the last sentence in
- By Peter Jolles
- No Comments
Guide to application of the Machinery Directive 2006/42/EC, Edition 2.1 – published in July 2017 and EN 61010-1 equipment
The Guide to application of the Machinery Directive 2006/42/EC, Edition 2.1, was published by the EC Commission in July. The link to the document
- By Peter Jolles
- No Comments
ATEX Directive 2014/34/EU and the ATEX Directive 99/92/EC
The ATEX Directive 2014/34/EU applies to equipment used in mines and potentially explosive atmospheres. It is very common for the sales staff at F2
- By Peter Jolles
- No Comments
Net Neutrality, what is it?
I have been hearing a lot about “net neutrality” lately but I did not really know what it means, probably the same as most
- By Peter Jolles
- No Comments
The Exemption for Medical Devices Under the RoHS Directive Has Expired
The RoHS Directive 2011/65/EU, Article 4 (3) exclusion for industrial monitoring and control instruments expired on Saturday, July 22, 2017. That exclusion was used
- By Peter Jolles
- No Comments
Safety Components and the Machinery Directive 2006/42/EC
From time to time we are presented with a device that seemingly “fits” into the Low Voltage Directive 2014/35/EU because it only has electric
- By Peter Jolles
- No Comments
CE marking and the ATEX Directive 2014/34/EU
Many times, we receive a request for assistance from a customer for a project that involves, “CE marking and ATEX.” While there is nothing
- By Peter Jolles
- No Comments
The EU declaration of conformity
A frequent source of confusion for exporters of products to the EU is the EU declaration of conformity. Many customers that we assist are
- By Peter Jolles
- No Comments
CE marking and the Technical File
Manufacturers who send equipment into the EU are required to send a CE compliant product in most cases. This means that if you are
- By Peter Jolles
- No Comments
Repeal of the Affordable Care Act and the Medical Device Tax
Yesterday afternoon, March 6th, the House Republicans released the bill to repeal (most of) the Affordable Care Act, also known as Obamacare. Since F2
- By Peter Jolles
- No Comments
Compliance Requirements for Lasers in the EU
Laser devices sent to Europe are subject to specific safety requirements. Some of these requirements are applicable even if the rest of the device
- By Peter Jolles
- No Comments
UL 94 Plastic Testing for Materials Used in Enclosures
UL 94 is a standard that measures, in a laboratory setting, the burn characteristics of plastic materials. The title of the standard itself is
- By Peter Jolles
- No Comments
The Machinery Directive 2006/42/EC and lifting accessories
The Machinery Directive 2006/42/EC, broadly, is applicable to machinery, defined in Article 2 (a) as an assembly fitted or intended to be fitted with
- By Peter Jolles
- No Comments
New RoHS restrictions, substances added to Annex II
There is a change on the horizon for manufacturers used to complying with RoHS based on the restriction of the six substances indicated in
- By Peter Jolles
- No Comments
Compliance with the Low Voltage Directive 2014/35/EU, Annex I
F2 Labs is constantly working to help customers better understand their responsibilities in complying with CE marking Directives that may or may not apply to
- By Peter Jolles
- No Comments
CE marking Directives and the Authorized European Representative
The CE marking is placed on products after compliance with all applicable EU CE marking Directives. Some typical examples of CE marking Directives that
- By Peter Jolles
- No Comments
Standards Used For Radio Product Legislation in the EU
The Radio Equipment Directive 2014/53/EU, or RED, came into force in June of this year. This new Directive replaced the Radio and Telecommunications Terminal
- By Peter Jolles
- No Comments
Primer For CE and other European Compliance Markings
Manufacturers who build equipment for sale to customers in the European Union may spend a lot of time learning about and understanding the process
- By Peter Jolles
- No Comments
Harmonized EN standards, CE marking Directives and your European projects
Silently and slowly – but inexorably… the Directives and standards we use for CE marking are updated, revised, and replaced – and we must
- By Peter Jolles
- No Comments
The Radio Equipment Directive and the Simplified EU Declaration of Conformity
I first read the new Radio Equipment Directive 2014/53/EU (aka the “RED”) in 2014 when it was published and noted its similarity to the
- By Peter Jolles
- No Comments
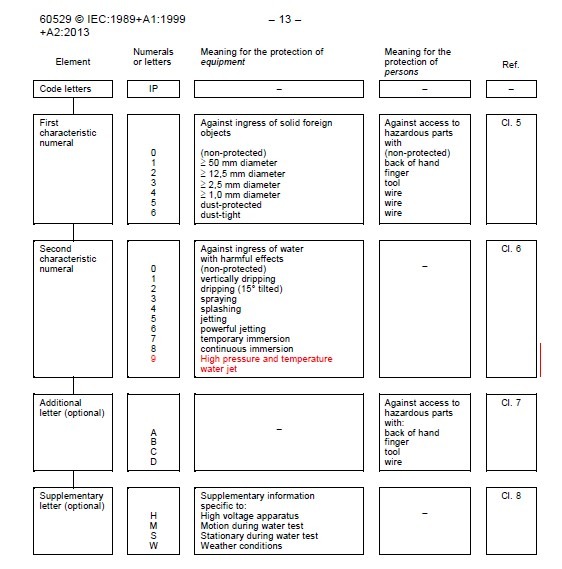
What is IEC 60529 IP Testing?
At F2 Labs, our IP testing chambers are constantly filled with projects undergoing testing to IEC 60529. Since I work in the sales department
- By Peter Jolles
- No Comments
Correct Labeling Requirements for the FCC
Recently the F2 Labs sales department was presented with a question from a call-in customer who wanted to know about the requirements from the
- By Peter Jolles
- No Comments
The Pressure Equipment Directive and machine builders
The Pressure Equipment Directive 97/23/EC was recast in 2014 and the new Pressure Equipment Directive 2014/68/EU replaced the old “PED” on July 19, 2016. This
- By Peter Jolles
- No Comments
CE marking and the China Export mark
There are two commonly used markings on equipment that are very similar in appearance but mean different things. The CE marking is a European
- By Peter Jolles
- No Comments
LED lighting and FCC requirements
Manufacturers of LED lighting sometimes assume that they are subject to FCC Part 18 requirements for lighting since LED’s are lights. Apparently this approach
- By Peter Jolles
- No Comments
The EMC Directive and “Fixed Installations”
A common discussion point with manufacturers is the notion of the “fixed installation”. Probably mostly due to recital (36) in the beginning of the
- By Peter Jolles
- No Comments
RoHS Directive 2011/65/EU – Equipment for Research and Development
The understanding of many CE marking Directives is enhanced oftentimes by comparing a specific product to the recitals, articles, and annexes of the Directive
- By Peter Jolles
- No Comments
The RoHS Directive 2011/65/EU and Correct Markings to Indicate Compliance
The RoHS Directive 2011/65/EU, or ‘RoHS2’ as it is sometimes called, is a CE marking Directive and is a European law. The various RoHS
- By Peter Jolles
- No Comments
Machinery Directive 2006/42/EC Article 1 (2) (k)
Previously we discussed some of the legal questions about compliance to either the Machinery Directive 2006/42/EC or the Low Voltage Directive 2014/35/EU. This one
- By Peter Jolles
- No Comments
Machinery Directive or the Low Voltage Directive… which one?
The Machinery Directive and the Low Voltage Directive: how do I know which one? The Machinery Directive 2006/42/EC and the Low Voltage Directive 2014/35/EU
- By Peter Jolles
- No Comments
CE marking and the Requirement for Native Language Translations
Your compliance requirements for export to the EU do not stop with putting the CE marking on your equipment. Depending upon the applicable CE